Intervention: Nutrafol Women’s Balance (n = 60).
Methods: This is an unblinded extension study, following subjects who were initially recruited to Ablon et al. (2021) At the end of the initial 6-month, placebo-controlled study, all remaining subjects entered this extended 6-month study. Subjects initially randomized to receive Nutrafol Women’s Balance continued with this supplement, and placebo-treated subjects from the original study were switched to Nutrafol Women’s Balance.
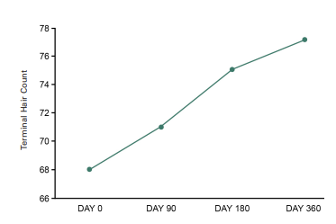
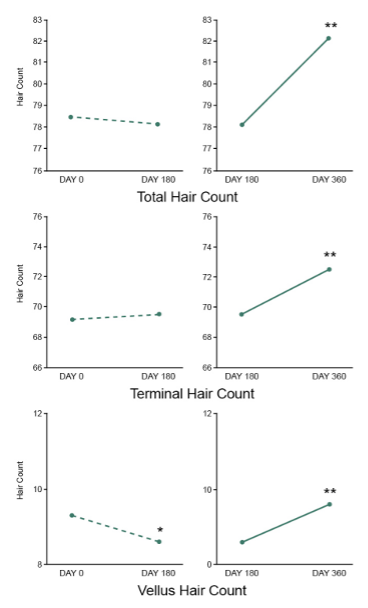
For this unblinded extension study, all subjects were additionally evaluated at Day 270 and Day 360. Terminal, vellus and total hair counts were assessed using phototrichograms. Hair shedding counts during hair washing, blinded physician global hair assessments for hair growth and quality, and responses on subjective self-assessments were also evaluated.
Study Duration: 12 months (6 months RCT + 6 months unblinded extension study)
Eligibility Criteria:
1. Females aged 40 – 65 who are perimenopausal, menopausal or post-menopausal.
2. In good health, as determined by responses to the initial study assessment.
3. Self-perceived hair thinning, confirmed on initial study assessment by the investigator.
4. Fitzpatrick I-IV photo skin types.
For more, plus the exclusion criteria, see:
ClinicalTrials.gov